In pulse electroplating, especially high-efficiency pulse plating with oxygen evolution and polarity reversal, insoluble anodes are subjected to severe operating conditions. Iridium may undergo complex valence changes, which accelerates the degradation rate of the precious metal coating. Meanwhile, the accelerated consumption of additives also presents a major challenge. Following the previous article on the mechanism of pulse electroplating, this paper discusses the electrochemical reactions occurring at electrodes under different systems. Taking iridium‑based tantalum‑titanium anodes for acidic bright copper plating as an example, the degradation mechanism of the precious metal coating on insoluble anodes is analyzed. Furthermore, high-performance insoluble anode solutions suitable for oxygen-evolving pulse copper plating are introduced, providing a reference for the industrial application of oxygen-evolving pulse copper plating processes.
To complete the entire circuit for electroplating, an anode is required in addition to the cathode. In metal electroplating, anodes are generally classified into soluble anodes and insoluble anodes. This discussion focuses on insoluble anodes.
In direct‑current (DC) electroplating, the general reaction at the cathode is:
Mn+ + ne⁻ = M
Side reactions at the cathode, such as hydrogen evolution, are not considered here.
In DC electroplating, the primary reaction at the insoluble anode is:
2H₂O = O₂↑ + 4H⁺ + 4e⁻
In pulse electroplating, especially reversing pulse electroplating, the anode undergoes the following reactions:
2H₂O = O₂↑ + 4H⁺ + 4e⁻(Forward pulse)
Mn+ + ne⁻ = M(Reverse pulse)
The reverse current density is typically about three times the forward current density.If the concentration of metal ions in the electrolyte is low, the following side reaction may occur during the reverse pulse:
2H⁺ + 2e⁻ = H₂↑(Reverse pulse)
For iron‑containing pulse systems, the electrochemical window of the anode is narrowed, and the dominant reactions become:
Fe²+ = Fe³⁺ + e⁻(Forward pulse)
Mn+ + ne⁻ = M(Reverse pulse)
Pulse systems characterized by oxygen evolution are generally referred to as oxygen‑evolution pulse systems, whereas those involving iron ions are called iron‑based pulse systems.
The above summarizes several possible reactions occurring on the surface of insoluble anodes in pulse systems.
We now turn to the insoluble anode itself.
Using acidic bright copper plating as an example, the primary insoluble anode currently employed is the iridium‑based tantalum‑titanium anode.
Starting with the precious metal iridium, the dominant form of iridium on this titanium anode is IrO₂ (iridium dioxide).
As an electrocatalyst for acidic water electrolysis, IrO₂ exhibits a low oxygen‑evolution overpotential and excellent acid resistance.
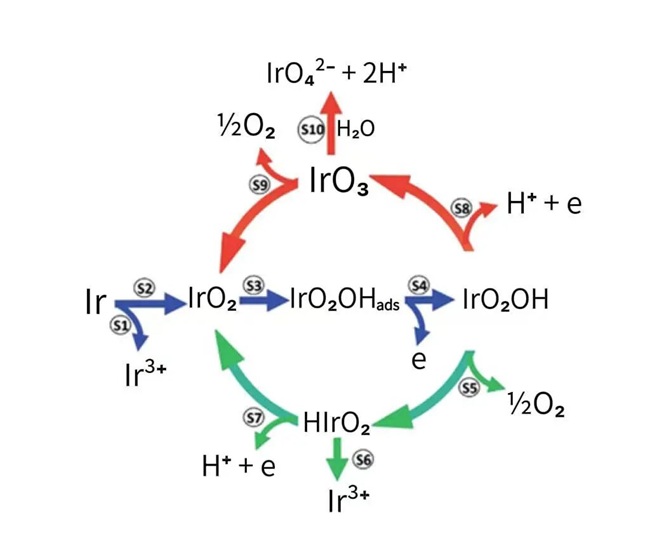
Figure 4-1 Cycling and Dissolution Mechanism of IrO₂ During Water Electrolysis
This figure presents a widely accepted view on the mechanism and degradation of IrO₂ during water electrolysis.
Among the species, Ir³⁺ is readily soluble in water, while IrO₃ reacts with water to form soluble compounds, although the kinetics of this reaction are very slow.
This water electrolysis cycle involves the repeated redox cycling of iridium among the oxidation states: Ir³⁺, Ir⁴⁺, Ir⁵⁺, and Ir⁶⁺.
This implies that even though the electrocatalytic material used in current insoluble anodes is IrO₂, iridium may undergo complex valence state changes under reversing pulse electroplating.
Particularly in oxygen‑evolving reversing pulse electroplating, the relatively wide electrochemical window of the anode can involve transitions among Ir³⁺, Ir⁴⁺, Ir⁵⁺, and Ir⁶⁺, resulting in relatively fast dissolution of iridium, which has been observed in laboratory experiments.
The iridium consumption rate follows the order below:
Oxygen‑evolving & hydrogen‑evolving reversing pulse >Oxygen‑evolving & copper‑depositing reversing pulse >Iron‑redox‑couple & copper‑depositing reversing pulse
This sequence corresponds exactly to the successive narrowing of the electrochemical window.
Furthermore, iridium consumption in oxygen‑evolving copper‑depositing systems is approximately 7–10 times faster than in conventional direct‑current (DC) electroplating.
Oxygen‑evolving reversing pulse operation results in a wider electrochemical window for the insoluble anode, which accelerates the consumption rate of precious metals.
Meanwhile, in PCB electroplating, the consumption of additives must also be considered, which further involves the structural design of insoluble anodes.
These two factors impose stringent challenges on the application of insoluble anodes in oxygen‑evolving reversing pulse electroplating.
In response, Magneto has innovatively developed a new generation of insoluble anodes for oxygen-evolving pulse copper plating lines for printed circuit boards (PCBs), providing a professional solution.

Magneto’s oxygen‑evolution pulse copper plating anodes feature the newly developed Selecta® MS‑6 coating, which effectively reduces additive consumption.We have also upgraded the coating stack structure and introduced an innovative interlayer concept.While ensuring outstanding baseline performance, these advancements further enhance the capabilities of the insoluble anode, enabling full compatibility with the industry’s top‑tier additive systems.The copper plating quality and anode performance fully meet customer requirements, greatly extending anode service life, significantly reducing replacement frequency and operating costs, and helping customers improve economic benefits while maintaining product quality.
Excellent service life performance
High compatibility with plating additives
Excellent through-hole plating capability
Significant cost advantages
Low additive consumption